-
Новости
- ИССЛЕДОВАТЬ
-
Страницы
-
Группы
-
Мероприятия
-
Reels
-
Статьи пользователей
-
Offers
-
Jobs
Decentralized Clinical Trials Market Growth Fueled by Patient-Centric Innovation
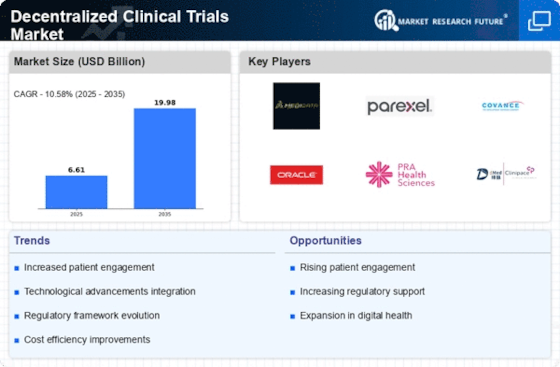
The Decentralized Clinical Trials Market Growth is strongly linked to the healthcare industry’s shift toward patient-centric research models. In traditional trials, participants often face logistical challenges such as long travel distances, frequent hospital visits, and time commitments that discourage participation. Decentralized clinical trials address these limitations by enabling remote data collection and virtual patient interactions. Through wearable devices, smartphone applications, and digital health platforms, researchers can continuously monitor patient health data in real time. This approach not only improves patient convenience but also enhances the accuracy of clinical data by capturing real-world health metrics outside controlled clinical environments. Pharmaceutical companies recognize the value of these capabilities, which help them gather richer datasets and improve trial outcomes.
Another factor accelerating decentralized clinical trial adoption is the increasing use of remote diagnostics and home-based testing kits. These technologies enable patients to complete many diagnostic procedures without visiting a medical facility. Healthcare providers can then review results through digital platforms and communicate with participants via telehealth consultations. Additionally, decentralized trials support more inclusive research by allowing participation from diverse geographic locations and demographic groups. This diversity leads to more representative clinical data and improves the effectiveness of new treatments across different populations. As the healthcare sector continues to prioritize digital transformation, decentralized clinical trials will play a vital role in shaping the future of pharmaceutical research and development.
FAQ
Q1. What is patient-centric clinical research?
Patient-centric research focuses on improving participant experience and accessibility during clinical trials.
Q2. How do wearable devices support decentralized trials?
Wearable devices collect continuous health data such as heart rate, activity levels, and sleep patterns.
Q3. Why is diversity important in clinical trials?
Diverse participation ensures treatments are safe and effective for broader populations.
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Игры
- Gardening
- Health
- Главная
- Literature
- Music
- Networking
- Другое
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


