Laboratory Developed Test Market Drivers: Rising Demand for Personalized Medicine and Advanced Diagnostics
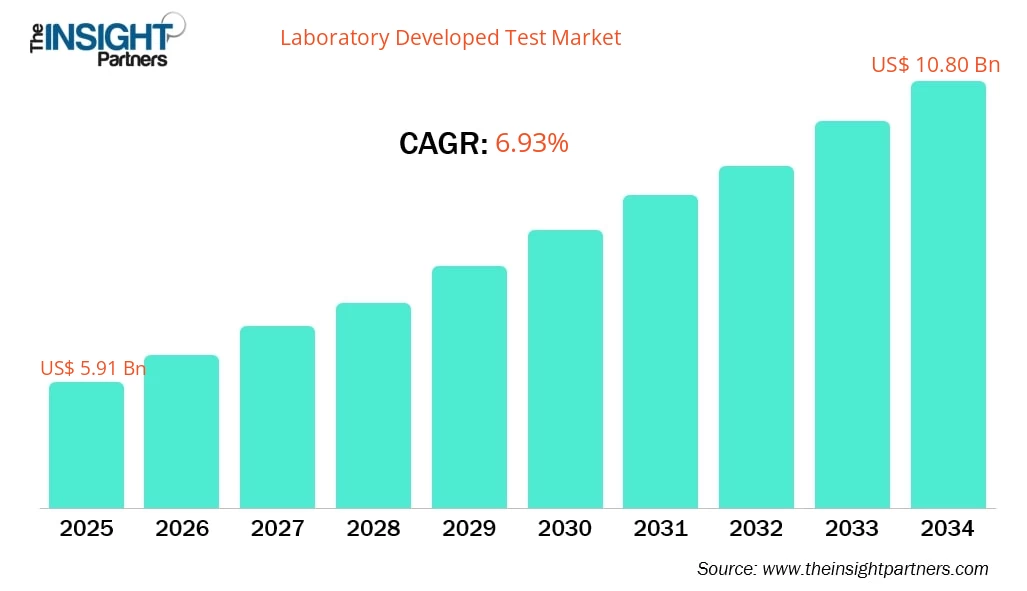
The Laboratory Developed Test (LDT) Market is gaining strong momentum as healthcare systems increasingly rely on advanced diagnostics to support precision medicine and early disease detection. The market is projected to grow from US$ 5.91 billion in 2025 to US$ 10.80 billion by 2034, registering a CAGR of 6.93% during 2026–2034. The expansion of molecular diagnostics, genomic research, and personalized healthcare continues to transform the global testing landscape. In this evolving ecosystem, Laboratory Developed Test (LDT) Market Drivers such as technological innovation, rising disease prevalence, and expanding healthcare infrastructure are accelerating the adoption of laboratory-developed tests worldwide.
Laboratory developed tests are in-house diagnostic assays designed, validated, and performed within a single laboratory. Their flexibility allows laboratories to rapidly create customized testing solutions for emerging clinical needs. LDTs play a critical role in oncology, infectious diseases, rare disease screening, and genetic testing. As healthcare providers demand faster, more accurate, and patient-specific diagnostics, the adoption of LDTs continues to expand across hospitals, research institutions, and specialized laboratories.
Download Sample PDF Copy:
https://www.theinsightpartners.com/sample/TIPRE00019603
Market Drivers
Rising Adoption of Precision Medicine
One of the most significant drivers of the LDT market is the global shift toward precision medicine. Healthcare systems are transitioning from traditional treatment approaches to personalized therapies based on genetic, molecular, and biomarker data. Laboratory developed tests enable clinicians to identify patient-specific disease characteristics, allowing targeted therapies and improved treatment outcomes. As precision medicine initiatives expand worldwide, laboratories are investing heavily in advanced testing capabilities, fueling the growth of the LDT market.
Increasing Burden of Chronic Diseases
The growing prevalence of chronic diseases such as cancer, cardiovascular disorders, diabetes, and autoimmune conditions is a major factor driving demand for specialized diagnostic testing. Early detection and continuous monitoring are critical for managing chronic illnesses, and LDTs offer laboratories the ability to create highly sensitive and specific tests tailored to these conditions. With aging populations and changing lifestyles contributing to rising disease incidence, the need for advanced diagnostics continues to increase.
Growth in Oncology Diagnostics
Cancer diagnostics represent one of the largest application areas for laboratory developed tests. LDTs are widely used for identifying tumor mutations, detecting biomarkers, and guiding targeted therapies. Advances in cancer genomics have enabled laboratories to develop tests that predict treatment response and monitor disease progression. As the global cancer burden continues to rise, the demand for personalized oncology diagnostics is expected to remain a key growth driver.
Expansion of Molecular Diagnostics and Genomic Technologies
Technological advancements in next-generation sequencing (NGS), polymerase chain reaction (PCR), and bioinformatics are transforming diagnostic capabilities. Modern laboratories can now analyze complex genetic data with high speed and accuracy. These innovations enable the development of sophisticated LDTs capable of detecting multiple biomarkers simultaneously. As the cost of genomic sequencing continues to decline, more healthcare providers are integrating molecular testing into routine clinical practice.
Growing Demand for Infectious Disease Testing
The importance of rapid and reliable infectious disease testing has increased significantly in recent years. Laboratories require flexible testing solutions that can quickly adapt to new and emerging pathogens. LDTs allow laboratories to develop assays for outbreak detection, surveillance, and response. The rising focus on global health preparedness and early disease detection continues to drive investment in advanced diagnostic solutions.
Expanding Healthcare Infrastructure in Emerging Markets
Emerging economies across Asia-Pacific, Latin America, and the Middle East are investing heavily in healthcare infrastructure and diagnostic capabilities. Growing awareness of preventive healthcare and rising healthcare spending are encouraging patients to seek early diagnostic testing. LDTs offer cost-effective and customizable solutions that can address region-specific disease patterns, making them highly attractive in developing healthcare markets.
Increasing Role of Automation and Digital Health
Automation and digital technologies are transforming laboratory workflows and improving diagnostic efficiency. Artificial intelligence and machine learning are being integrated into laboratory processes to enhance accuracy, streamline operations, and reduce turnaround times. Automated systems allow laboratories to process large volumes of tests efficiently, supporting the growing demand for diagnostic services. These technological advancements are strengthening the adoption of LDTs globally.
Favorable Reimbursement and Healthcare Spending Trends
Healthcare payers and governments are increasingly recognizing the value of early diagnosis and personalized treatment in reducing long-term healthcare costs. Favorable reimbursement policies and rising healthcare expenditure are encouraging laboratories to develop and adopt advanced diagnostic tests. As reimbursement frameworks continue to evolve, laboratories gain greater financial support for innovation and expansion.
Competitive Landscape
The Laboratory Developed Test Market is highly competitive, with leading companies investing in research, collaborations, and product development to strengthen their market positions. Key players include:
- F. Hoffmann-La Roche Ltd.
- Thermo Fisher Scientific Inc.
- Illumina, Inc.
- QIAGEN N.V.
- Agilent Technologies, Inc.
- Bio-Rad Laboratories, Inc.
- Danaher Corporation
- PerkinElmer, Inc.
- Quest Diagnostics Incorporated
- Laboratory Corporation of America Holdings
These organizations are focusing on technological advancements, strategic partnerships, and expanding diagnostic portfolios to meet the growing demand for advanced testing solutions.
Future Outlook
The Laboratory Developed Test Market is expected to witness steady growth throughout the forecast period. The convergence of genomics, digital health, and personalized medicine will continue to create new opportunities for innovation. As laboratories enhance their capabilities and healthcare systems prioritize early diagnosis, LDTs will remain essential for improving patient outcomes and supporting efficient clinical decision-making.
Related Reports –
In Vitro Diagnostics (IVD) And Laboratory Developed Tests For Autoimmune Diseases Market
Laboratory Thermostat Market Growth, Demand & Size by 2034
Laboratory Cabinets Market Growth, Share & Trends by 2034
About Us:
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services. We specialize in industries such as Semiconductor and Electronics, Aerospace and Defense, Automotive and Transportation, Biotechnology, Healthcare IT, Manufacturing and Construction, Medical Device, Technology, Media and Telecommunications, Chemicals and Materials.
Contact Us:
Ankit Mathur | The Insight Partners
E-mail: ankit.mathur@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in : Korean | German | Japanese | French | Chinese | Italian | Spanish
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jogos
- Gardening
- Health
- Início
- Literature
- Music
- Networking
- Outro
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


